Supply chain resilience is a critical challenge in the life sciences industry, where manufacturing disruptions can take years to resolve and directly impact patient care. The complexity of regulatory compliance, combined with long lead times for scaling production, makes proactive planning and risk management essential.
Success requires deep operational expertise to build resilient supply chains that can consistently deliver critical treatments to patients. A pharmaceutical company engaged L.E.K. Consulting’s Operations and Supply Chain team to help address these challenges.
Tackling the resilience challenge
While all pharmaceutical companies must navigate complex manufacturing and regulatory requirements, the company’s siloed structure — managing drug substance, product and packaging independently — further complicated its situation.
As pharmaceutical pipelines grow more complex, with increasingly specialized treatments and global regulatory requirements, managing supply chains will only become more challenging. Rising costs and shifting market dynamics further underscore the need for proactive resilience strategies. For this company, several structural constraints made it even more difficult to build the necessary resilience into its supply chain:
- Manufacturing complexity: Contract development and manufacturing organizations impose high minimum orders and limited production flexibility.
- Qualification time: Adding production lines or suppliers requires 12-18 months of validation.
- Inventory costs: Safety stock for specialized products can reach tens of thousands of dollars per unit.
- Regulatory burden: Process or facility changes need extensive documentation and approval.
L.E.K.’s approach began with forming cross-functional teams for each product, conducting baseline assessments to align understanding as well as training teams on how changes affect upstream and downstream operations.
Mapping and modeling the supply chain
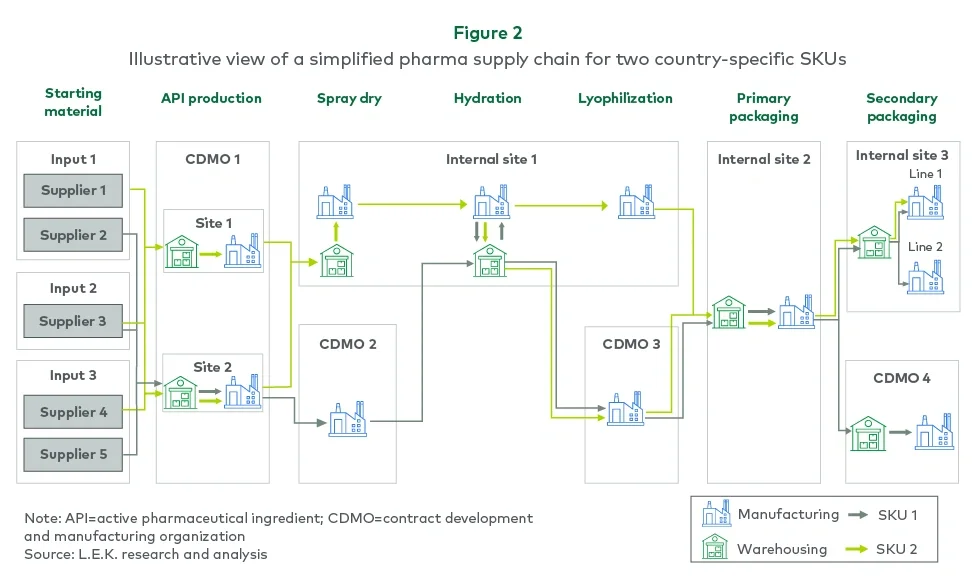
Detailed supply chain mapping revealed hidden vulnerabilities in the company’s siloed operations. Working backward from finished goods, the L.E.K. team created a digital representation showing how demand translated into capacity requirements.
This process uncovered critical constraints — some active pharmaceutical ingredient (API) production suites could only produce certain country-specific SKUs, while certain sterile fill-finish lines were restricted to specific API materials. These limitations showed how overlooked details could compromise resilience during disruptions.
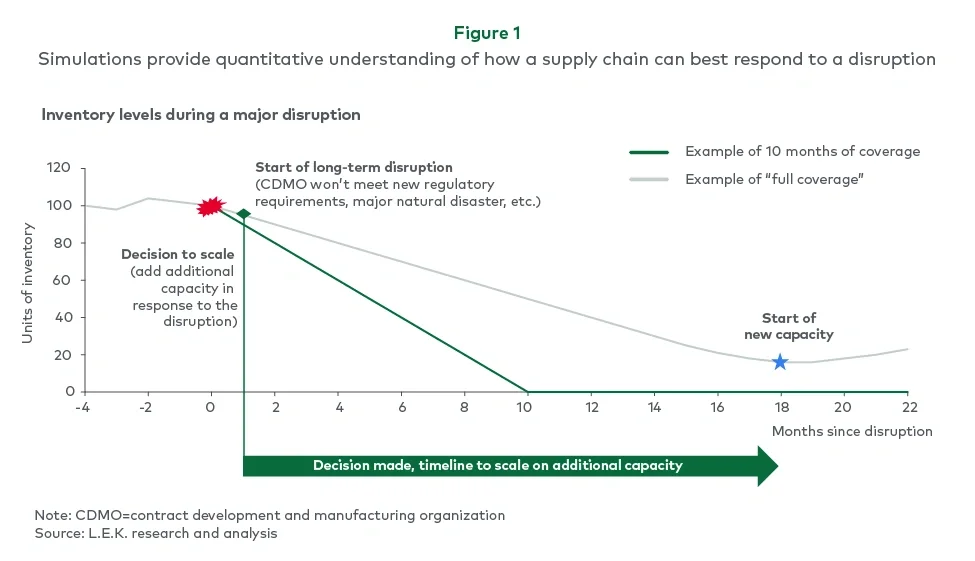
During the engagement, a major disruption at a critical facility provided a real-world test of the supply chain’s resilience. The event threatened to halt production of multiple products, demonstrating how a single point of failure could quickly cascade throughout the network.
This concrete example helped reinforce executive buy-in for implementing more robust contingency measures and extended coverage across the portfolio (see Figure 1).